Confidently rule out melanoma with the DermTech Melanoma Test® 1
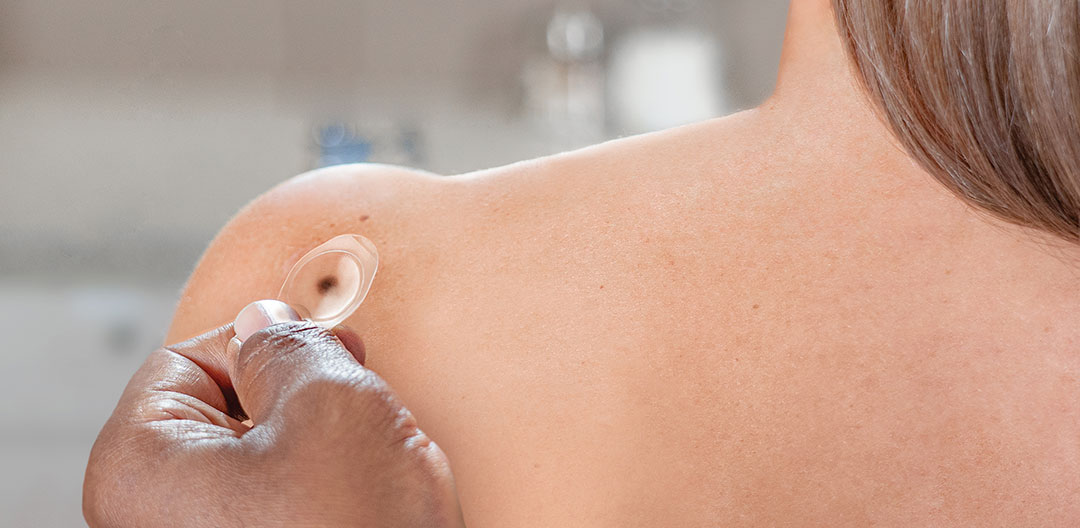
Ask your Dermatologist about the DermTech Melanoma Test.
Recommended use of the DermTech Melanoma Test
The DermTech Melanoma Test is indicated for use on pigmented lesions that:
- Meet one or more ABCDE criteria
- Have a diameter of ≥ 5mm
- Are on patients 18 years or older
How to use the DermTech Melanoma Test
Learn how to use the DermTech Smart Stickers to collect skin cells from an equivocal pigmented lesion. It takes less than 5 minutes per lesion and can be performed by medical staff or at home under supervision.
Adopt the DermTech Melanoma Test into your practice today.
aPlease refer to LCD #38151 for additional Medicare coverage criteria.
References:
1. Gerami P, et al. J Am Acad Dermatol. 2017;76(1):114-120. 2. Jackson S, et al. SKIN J Cutan Med. 2020;4(2):105-110. 3. Ferris L, et al. Dermatol Online J. 2019;25(5):2.

